Division of Environmental Quality Director: Art Goodin
Mercury released outdoors is a problem. Natural sources of mercury releases to the environment include volcanic eruptions and forest fires. In addition to natural sources, there have been decades of mercury releases from the burning of coal, the production of paper and chlorine, waste incineration and other industrial processes. Because it is an element, mercury does not break down into less harmful substances. It will circulate in the atmosphere until it falls to earth attached to rain or dust particles. Eventually it makes its way to lakes and streams where bacteria transform it into methylmercury, a much more toxic form. In an aquatic environment, mercury reaches its highest concentrations in the larger predatory fish - the type most often sought by anglers.
Mercury in Fish
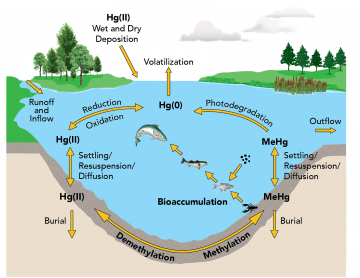
The majority of mercury in the environment is in a gaseous form, shown here as elemental mercury or Hg(0). In the atmosphere, elemental mercury is converted to a more water-soluble form, ionic mercury or Hg(II), which is returned to the earth's surface in precipitation. Bacteria can convert these forms of mercury to methylmercury (MeHg), a much more toxic form that can concentrate quickly up the food chain. The amount of mercury in the air, plants, animals, land and water is constantly cycling between each of these components.
Organic mercury compounds are formed when elemental mercury combines with carbon. When mercury is released to the environment some of it gets converted by microscopic organisms into methylmercury, a highly toxic organic mercury compound. Methylmercury is highly soluble, mobile and bioaccumulative in the food chain. Bioaccumulation is the process where organisms take up a contaminant faster than their bodies can eliminate it. Methylmercury can move quickly from water to sediment, aquatic plants, invertebrates and fish. It concentrates, or biomagnifies, as it moves up through the food chain. Biomagnification is the incremental increase in a contaminant's concentration at each level of the food chain. In an aquatic environment, mercury reaches its highest concentrations in the larger predatory fish - the type most often sought by anglers.
All fish contain small amounts of mercury. For most people, the health benefits of eating fish greatly outweigh the potential risks. Fish are an important source of protein, omega-3 fatty acids and other key nutrients that help maintain cardiovascular health. However, there are occasions when limited consumption of certain fish is appropriate. For example, sensitive populations—women who are pregnant or may become pregnant, nursing mothers and children younger than 13 years—are recommended to limit consumption of certain fish to reduce mercury exposure. These groups should eat no more than one meal per month of large predatory fish greater than 12 inches in length. Additional consumption recommendations can be found on the Missouri Department of Health and Senior Services Missouri Fish Advisory webpage This advisory is not intended to discourage anglers from eating fish, but should be used as a guide to eating fish low in contaminants.
In addition to Missouri's advisories, the U.S. Food and Drug Administration and Environmental Protection Agency have issued a saltwater fish consumption advisory for women of childbearing age who are pregnant or may become pregnant, nursing mothers and young children. The FDA recommends these individuals should not eat shark, swordfish, king mackerel and tilefish, and should limit consumption of shrimp, canned light tuna, salmon, pollock and catfish. Albacore tuna has higher levels of mercury than other tuna and so greater limitations on consumption of albacore tuna are also recommended.
Learn More
- Fish Tissue Monitoring
- Guidelines for Eating Fish that Contain Mercury - U.S. Environmental Protection Agency
- Mercury in Missouri Streams and Lakes - PUB2100
- Missouri Fish Advisory - Missouri Department of Health and Senior Services
Nothing in this document may be used to implement any enforcement action or levy any penalty unless promulgated by rule under chapter 536 or authorized by statute.
For more information
Division of Environmental Quality
1101 Riverside Drive
P.O. Box 176
Jefferson City, MO 65102-0176
United States